Rufen Sie uns einfach an, und wir beraten Sie gerne zu unserem Seminar- und Studienangebot.
Unsere Ansprechpartner:

Michael Rabbat, Dipl.-Kfm.
MBA Chief Operating Officer

Claudia Hardmeier
Kunden-Center
Studienbetreuung
6.2. Stepwise roll out of a new concept
After full approval from top management implementation can be initiated, a consultant needs to be identified and internal champions be nominated. The core team then starts the roll out according to the milestone plan and reports back to management.
Overcoming the hurdle of widespread acceptance by training of all project teams first requires the definition of a corporate process for a QbD driven development approach. This is the most challenging task for the team to achieve at the start. Though the principles of such an approach are clearly understood the adaptation and translation of the work flow from ICH Q8(R2) into actual processes within an organisation with existing, traditional systems proofs to be a challenging task with many interfaces to consider.
As such the need for a development handbook for QbD becomes more important than initially envisioned. The core parts of this are definitions of the terminology, the process flow depicted in Figure 12 and a list of additional QbD documents needed.

Figure 12: QbD process at Pharma Example Ltd.66
This process actually builds a continuous cycle throughout development, starting with the initial definition of a target profile, based on which the initial risk evaluation is made. Thereby, the potential critical attributes are identified and then the material attributes and process parameters that impact these essential product attributes and are thus considered to be critical can be determined. Based on this risk assessment and the potential criticality of individual aspects, experimental investigations and actions are initiated to determine the actual criticality and mitigate critical risks as good as possible by the chosen control strategy and/or further investigations. This reciprocating cycle will continue throughout development, increasing the knowledge on the drug product and the manufacturing process and thus the validity of the control strategy and the resulting quality. The established Design Space and the established Knowledge Management will then foster Continual Improvement throughout the product’s lifecycle.
Table 5 describes the documents and their contents, when to generate these, who needs to initiate a document, who needs to write, contribute, review and authorise these documents, when to update them, how to archive and who to inform about their contents. Thus this list in the development handbook for QbD describes not only the responsibilities and accountabilities for these documents, but also the required processes behind them to live the QbD cycle as depicted in Figure 12. These processes can thus act as surrogates for yet missing integrated management processes and procedures, which may be implemented in the organisation in parallel, in cooperation with other line or matrix functions.
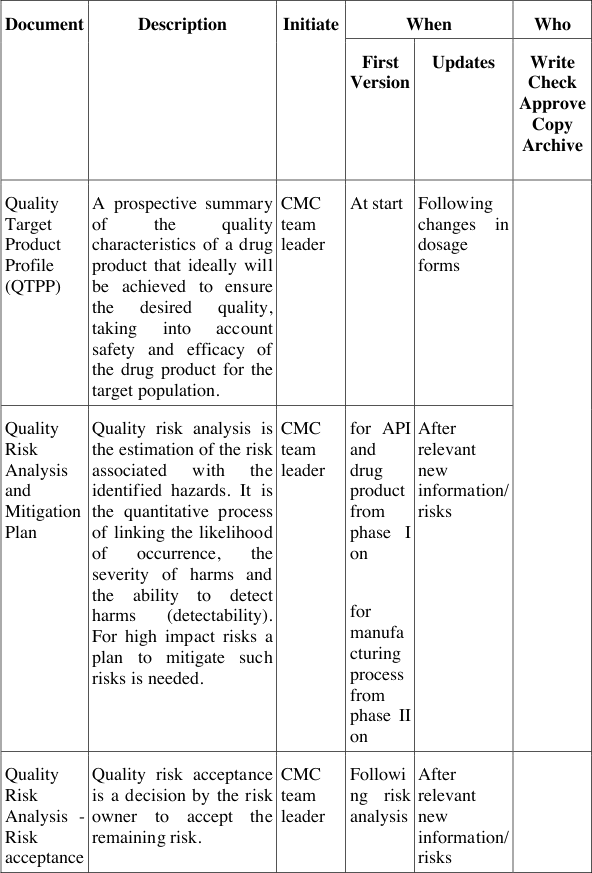
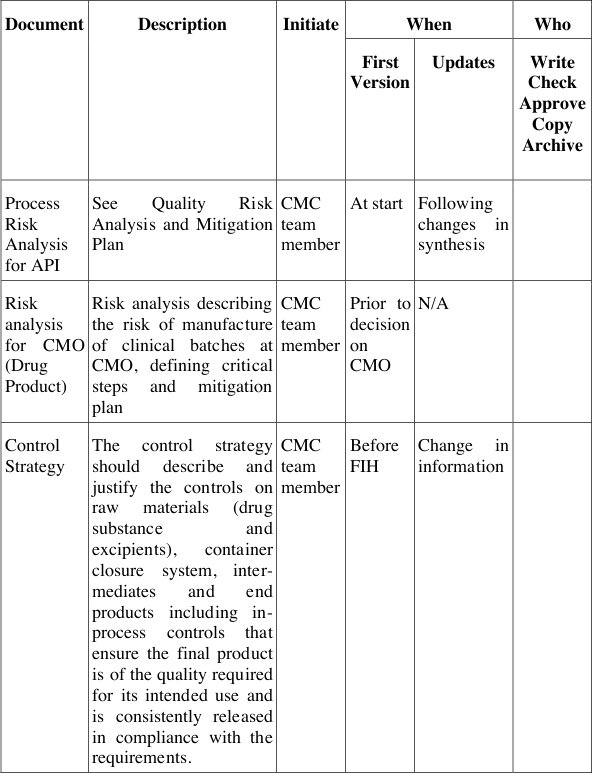
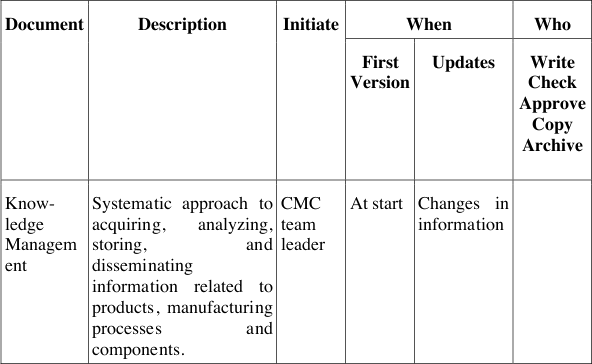
Table 5: QbD documents and responsibilities at Pharma Example Ltd.
The QbD documents generated during development ensure the systematic approach in each project and also the comparability of projects when evaluating the risks of a project and the extent of mitigation needed. The extent of activities and investigations, however, is not pre-defined but can be determined by each project team according to its willingness and strategic decisions to accept certain risks at a certain stage or not. By documenting both, the risk assessment and the risk acceptance these decisions can always be tracked and re-evaluated over the course of a project if needed.
Together with the objective to focus on really critical risks rather than following a rigid scheme of investigations and experimentation for each phase, this approach combines the quality driven, systematic development approach of QbD with the Minimalism approach of doing the relevant tests to identify between systematic and unsystematic errors and to differentiate how to approach each of these adequately. Risk analysis, assessment and acceptance therefore provide the linking tool for using the other tools of QbD effectively and efficiently, without jeopardizing critical product quality and product performance in the volunteer or patient at any time.
Thereby the number of investigations needed at each phase of development can actually be decreased, depending on the criticality of an active compound, drug product or manufacturing process while still addressing all risks adequately at the most suitable timing for the project to successfully improve the quality for the customer at reduced timelines and costs and increased probability of success for a new drug product.
66 Source: author







